Hydrogen Reduction Lab

As a metallurgical research group, we specialize in high-temperature reduction processes:
“We discover and define the scientific fundamentals essential for a successful industrial transformation towards sustainable production.”
Our research focuses on material behavior under high-temperature conditions, especially during hydrogen-based reduction processes. Our approach integrates precise thermodynamic measurements, detailed kinetic experiments, and advanced microscopy to evaluate reaction behavior and microstructural evolution. By identifying reaction pathways, determining rate-limiting steps, and clarifying how specific process parameters affect reaction efficiency and material properties, we provide a robust scientific basis for optimizing processes at an industrial scale.
Publications with corresponding authorship
Towards Circular Steelmaking: Hydrogen-Based Valorization of Electric Arc Furnace Dust
Hydrogen reduction of EAF dust achieves near-quantitative zinc recovery (≈99%), higher iron metallization and markedly better S/P removal than carbothermic routes. The improved performance stems from H₂-driven kinetics that avoid pore closure, enabling even selective zinc extraction—supporting circular, lower‑carbon steelmaking.

Impact of temperature and gas composition on hydrogen-based zinc recovery from electric arc furnace dust
Hydrogen-enabled zinc recovery from electric arc furnace dust requires a narrow thermal window near 1150 °C and tuned H₂/H₂O ratios to balance volatilization and sintering. The study demonstrates high zinc extraction with preserved particle morphology, outlining a low-carbon pathway to recyclable zinc concentrate and secondary-grade DRI.
Comparison of reduction kinetics of Fe2O3, ZnO-Fe2O3 and ZnO with hydrogen (H2) and carbon monoxide (CO)
This study provides a detailed comparison of hydrogen (H2) and carbon monoxide (CO) reduction kinetics for the principal oxides in electric arc furnace dust: Fe2O3, ZnOFe2O3, and ZnO. Hydrogen delivers faster kinetics—about 2.5 times for Fe2O3, twice for ZnOFe2O3, and 1.5 times for ZnO—while revealing diffusion-controlled behaviour for Fe2O3 and chemically controlled kinetics for ZnO. The findings emphasise the influence of gas flow and temperature on hydrogen-based reduction, underscoring its potential for efficient, sustainable EAFD recycling.
The catalytic effect of the metal bath on the zinc oxide reduction
Catalytic reduction of zinc oxide using carbon dissolved in metal baths significantly accelerates zinc recovery from electric arc furnace dust. The study quantitatively demonstrates that carbon-saturated nickel enhances reaction kinetics by a factor of 10, while carbon-saturated iron achieves up to 30-fold acceleration, attributed to both carbothermic and metallothermic reactions. These insights outline an efficient, catalytic pathway toward improved recycling processes and economic zinc recovery.
A review on the fundamentals of hydrogen-based reduction and recycling concepts for electric arc furnace dust extended by a novel conceptualization
Hydrogen-based reduction offers a promising low-carbon alternative for recycling electric arc furnace dust (EAFD), a significant waste stream from steel production. This review systematically explores thermodynamic and kinetic aspects, illustrating that hydrogen facilitates faster reaction rates compared to carbon-based methods. Existing and novel process concepts are categorised, highlighting options for complete and selective reduction of zinc and iron oxides. A newly proposed concept significantly minimises hydrogen consumption by selectively reducing zinc oxide and recovering hydrogen through reoxidation, paving the way for economically viable and sustainable EAFD recycling solutions.
Carbothermic Reduction of Zinc Containing Industrial Wastes: A Kinetic Model
Carbothermic reduction is still state-of-the-art for recycling zinc-rich industrial wastes like electric arc furnace dust (EAFD). This study develops a kinetic model for the simultaneous carbothermic and metallothermic reduction of ZnO in a molten metal bath, showing metallothermic reactions contribute significantly (25-50%) to Zn recovery. Experiments confirm rapid kinetics and over 99.9% Zn recovery at 1400-1500 °C, supporting efficient industrial recycling of EAFD.
Innovation
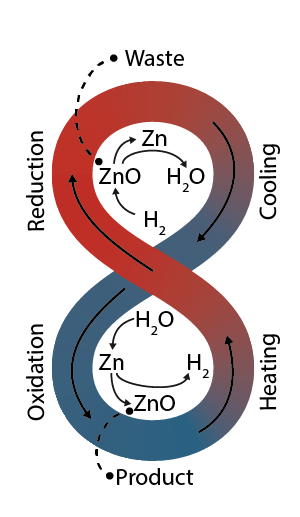
A method and plant for recycling metal oxides
This patent application introduces an innovative recycling method specifically developed for efficiently recovering zinc oxide from electric arc furnace (EAF) dust and similar metal oxide wastes. Central to the invention is a sealed rotary kiln utilizing a hydrogen-rich gas atmosphere that directly reduces metal oxides at precisely controlled high temperatures. Notably, zinc vapor formed during reduction is intentionally reoxidized using steam, regenerating hydrogen gas and capturing heat, both of which are recirculated back into the process. This unique closed-loop design substantially reduces energy consumption and emissions, distinguishing it significantly from conventional carbon-based recycling methods. Additionally, the process generates direct-reduced iron (DRI) as a valuable coproduct, enhancing economic viability and transforming hazardous waste streams into sustainable resources.
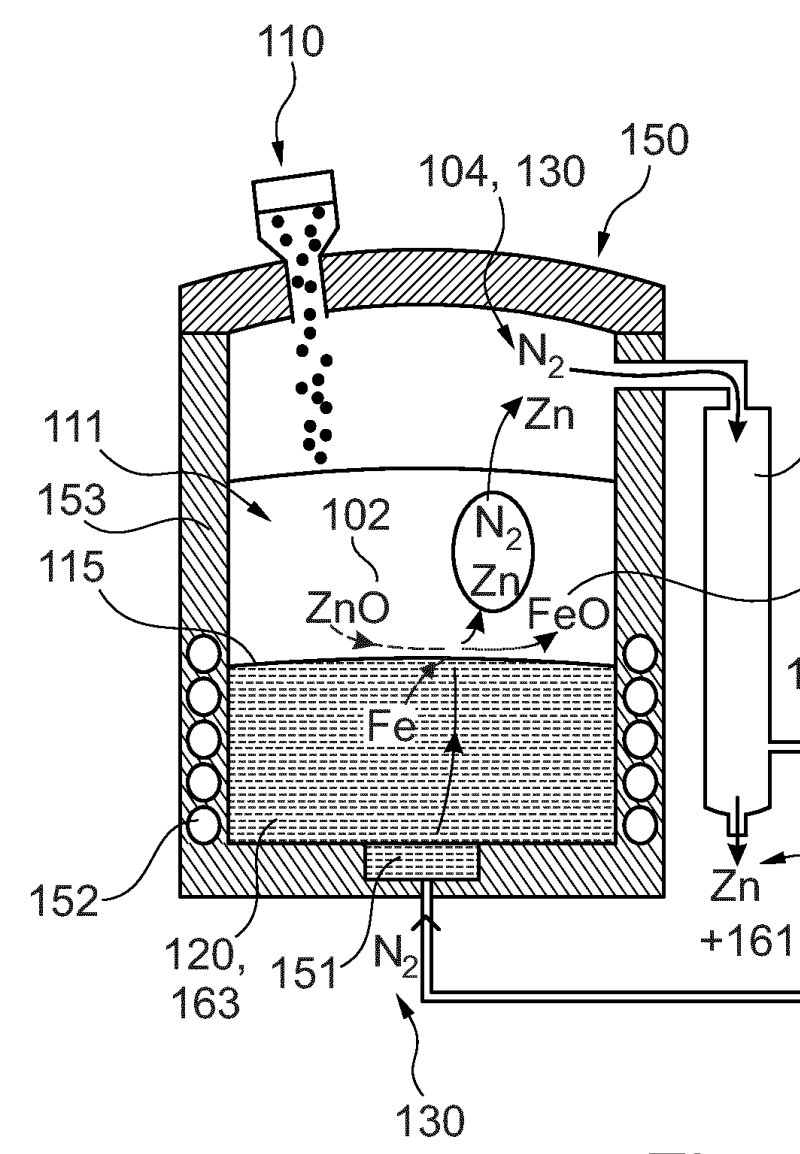
A method for separating zinc from an industry product using a pyrometallurgical reaction
This patent application describe a novel pyrometallurgical method designed to efficiently recover metallic zinc directly from electric arc furnace dust. Our kinetic studies found a surprisingly rapid reaction between zinc oxide (ZnO) and metallic iron. Based on these findings, we've developed a process that avoids carbon and therefore prevents the formation of gaseous by-products like CO2 or H2O. The result is simplified zinc condensation step in inert atmosphere without the usual reoxidation issues. The direct recovery of metallic zinc instead of zinc oxide improves the overall energy efficiency. Unlike the conventional Waelz kiln this method directly produces metallic zinc. Moreover, the it inherently separates additional valuable metals like copper and nickel, turning a waste stream into a genuine multi-metal recycling opportunity.
Ongoing Projects

Dust2Value
Pioneering Sustainable Recovery in Steelmaking: Hydrogen-Based Technology for Byproduct Management
Electric Arc Furnace Dust (EAFD) is a hazardous waste generated during steel production, containing significant amounts of zinc that must be recycled due to environmental and economic reasons. Currently, the standard recycling method is the Waelz kiln process, which recovers zinc in the form of secondary zinc concentrate and yields a stable, non-hazardous slag. However, this conventional approach has substantial environmental drawbacks, particularly high CO2 emissions. The Dust2Value project addresses these sustainability issues by scaling up an innovative hydrogen-based recycling technology. Unlike traditional methods, this process directly reduces zinc oxide to zinc vapor within a controlled atmosphere, dramatically lowering CO2 emissions. In addition, it yields a high-quality zinc concentrate and produces reusable direct reduced iron (DRI), providing a significantly more sustainable, efficient, and scalable alternative.
Role: Coordinator/Project Manager
Team

Dr. Manuel Leuchtenmüller
Team Lead
Dr. Manuel Leuchtenmüller is a metallurgical engineer specializing in hydrogen-based high-temperature processes and metal reduction kinetics. His research combines advanced thermodynamic measurements, kinetic modeling, and precise microstructural analysis to unravel reaction pathways and identify critical rate-limiting mechanisms in metal recycling processes. Through innovative coupling of thermogravimetric analysis and microscopy techniques, Manuel bridges fundamental insights with industrial-scale solutions. He actively translates scientific knowledge into practical, low-carbon metallurgy applications, emphasizing sustainable metal recovery from industrial waste. Currently, he coordinates the Horizon Europe Dust2Value project, leading a consortium dedicated to developing hydrogen-driven recycling technologies for zinc and iron recovery from steelmaking residues.
manuel.leuchtenmuller@unileoben.ac.at
Aaron
PhD Researcher
Develops algorithms, embedded hardware, and machine learning models for analyzing reduction kinetics in thermogravimetric systems. His work focuses on real-time data acquisition and subsequent interpretation to improve accuracy and process understanding in hydrogen-based reduction experiments.

Aleksandra
PhD Researcher
Specializes in process and reaction engineering of partially reduced EAF dusts. Her research links microstructural evolution to leaching behavior, combining hydrogen reduction studies with advanced SEM characterization to optimize integrated pyrometallurgical–hydrometallurgical recovery routes.
Student Researchers
Our student team powers day-to-day experiments, data wrangling, and visualization development.

Sofia
Data Evaluation

Markus
Thermogravimetric Analysis

Melanie
Heating Microscope

Mathias
Thermogravimetric Analysis

Theresa
Floation
Specialized in-house techniques
Thermogravimetric analysis coupled with mass spectrometry (TGA-MS) enables highly reproducible kinetic experiments under precisely controlled gas atmospheres for temperature- and gas-programmed reduction studies. Scanning electron microscopy (SEM) with multi-detector elemental mapping delivers phase identification, trace-element detection, porosity assessment, and phase-specific grain size distributions for feed and product characterisation. Heating microscope measurements capture melting onset, shape evolution, and structural change in real time, providing actionable inputs for furnace design, feed conditioning, and sintering control.

Research at Nonferrous Metallurgy

The X-MAT team investigates the behaviour of metals and alloys under extreme conditions — such as high temperatures, irradiation, hydrogen gas, plasmas, and corrosive media — relevant to space, fusion energy, hydrogen infrastructure, and harsh industrial settings.
Visit x-mat.unileoben.ac.at